
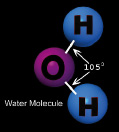
 The unique property of the water molecule is its shape.
It consists of one oxygen atom and two hydrogen atoms. You would
normally think that the water molecule would have an oxygen atom in the
middle with the hydrogen atoms on opposite sides. Because of the design
of the oxygen atom, that is not the way the molecule is arranged. The
oxygen atom only allows bonding to hydrogen in positions that are 90
degrees away from each other, so the hydrogen atoms end up being close
enough to one another to repel each other making the angle between them
105 degrees. This configuration gives the water molecule a polarity,
with the hydrogen side being positively charged and the opposite side
being negatively charged. Adjacent water molecules can be attracted to
these charged areas, giving water its unusual properties.
The unique property of the water molecule is its shape.
It consists of one oxygen atom and two hydrogen atoms. You would
normally think that the water molecule would have an oxygen atom in the
middle with the hydrogen atoms on opposite sides. Because of the design
of the oxygen atom, that is not the way the molecule is arranged. The
oxygen atom only allows bonding to hydrogen in positions that are 90
degrees away from each other, so the hydrogen atoms end up being close
enough to one another to repel each other making the angle between them
105 degrees. This configuration gives the water molecule a polarity,
with the hydrogen side being positively charged and the opposite side
being negatively charged. Adjacent water molecules can be attracted to
these charged areas, giving water its unusual properties.
In Science News (January
26, 2008, page 58) researchers report new studies on what happens to
water as it continues to cool and is put under pressure. The  title
of
the
article is "Supercool, and Strange." The subtitle is "Scientists
are finding clues about why water is so utterly weird." In the article
is a reference to a Web site where water's anomalies are listed. It is
http://www.lsbu.ac.uk/water/anmlies.html. The site has numerous
references and explanations, many of which are quite technical. Here is
a portion that is not too technical and that will give you an idea of
the strange properties of water.
title
of
the
article is "Supercool, and Strange." The subtitle is "Scientists
are finding clues about why water is so utterly weird." In the article
is a reference to a Web site where water's anomalies are listed. It is
http://www.lsbu.ac.uk/water/anmlies.html. The site has numerous
references and explanations, many of which are quite technical. Here is
a portion that is not too technical and that will give you an idea of
the strange properties of water.
Although it is an apparently simple molecule, (H2O), it has a highly complex and anomalous character. As a gas, water is one of lightest known, as a liquid it is much denser than expected and as a solid it is much lighter than expected when compared with its liquid form.As liquid water is so common-place in our everyday lives, it is often regarded as a "typical" liquid. In reality, water is most atypical as a liquid, behaving as a quite different material at low temperatures to that when it is hot. It has often been stated ... that life depends on these anomalous properties of water. In particular, the high cohesion between molecules gives it a high freezing and melting point, such that we and our planet are bathed in liquid water. The large heat capacity, high thermal conductivity and high water content in organisms contribute to thermal regulation and prevent local temperature fluctuations, thus allowing us to more easily control our body temperature. The high latent heat of evaporation gives resistance to dehydration and considerable evaporative cooling. Water is an excellent solvent due to its polarity, high dielectric constant and small size, particularly for polar and ionic compounds and salts. It has unique hydration properties towards biological macromolecules (particularly proteins and nucleic acids) that determine their three-dimensional structures, and hence their functions, in solution. This hydration forms gels that can reversibly undergo the gel-sol phase transitions that underlie many cellular mechanisms. Water ionizes and allows easy proton exchange between molecules, so contributing to the richness of the ionic interactions in biology.
At 4°C water expands on heating or cooling. This density maximum together with the low ice density results in (i) the necessity that all of a body of fresh water (not just its surface) is close to 4°C before any freezing can occur, (ii) the freezing of rivers, lakes and oceans is from the top down, so permitting survival of the bottom ecology, insulating the water from further freezing, reflecting back sunlight into space and allowing rapid thawing, and (iii) density driven thermal convection causing seasonal mixing in deeper temperate waters carrying life-providing oxygen into the depths. The large heat capacity of the oceans and seas allows them to act as heat reservoirs such that sea temperatures vary only a third as much as land temperatures and so moderate our climate (for example, the Gulf stream carries tropical warmth to northwestern Europe). The compressibility of water reduces the sea level by about 40 m giving us 5% more land. Water's high surface tension plus its expansion on freezing encourages the erosion of rocks to give soil for our agriculture.
Notable amongst the anomalies of water are the opposite properties of hot and cold water, with the anomalous behavior more accentuated at low temperatures where the properties of supercooled water often diverge from those of hexagonal ice. As (supercooled) cold liquid water is heated it shrinks, it becomes less easy to compress, its refractive index increases, the speed of sound within it increases, gases become less soluble and it is easier to heat and conducts heat better. In contrast as hot liquid water is heated it expands, it becomes easier to compress, its refractive index reduces, the speed of sound within it decreases, gases become more soluble and it is harder to heat and a poorer conductor of heat. With increasing pressure, cold water molecules move faster but hot water molecules move slower. Hot water freezes faster than cold water and ice melts when compressed except at high pressures when liquid water freezes when compressed. No other material is commonly found as solid, liquid and gas.
(from "Water Structure and Science" by Martin Chaplin)
The author then lists 64 unique properties of water. This is a technical discussion aimed at people with a background in science, and we encourage readers with science backgrounds to read through this incredibly interesting material.
The unique design of the water molecule that allows all of this strange behavior is vital to our own physical existence. This is a powerful demonstration of the fact that things as simple as water are not as simple as they first appear. It speaks eloquently of the intelligence that created the cosmos in which we live.
Back to Contents
Does God Exist?, JanFeb09.
11/23/2010